How to Read an Autopsy Toxicology Report Brain
Introduction
Opioid overdose deaths go on to increase in the United States, killing more than 42,000 people in 2016. The opioids detected in these cases, in increasing order, were methadone, natural and semi-constructed opioids (east.g., oxycodone, hydrocodone), heroin and synthetic opioids (e.g., fentanyl, fentanyl-analogs). Synthetic opioids (excluding methadone) and heroin deaths specifically experienced a sharp increase from 2015 to 2016 (20 and 100%, respectively) (Seth et al., 2018). Fentanyl and its derivatives take been increasingly present every bit adulterants mainly in heroin, simply also in other drugs such as cocaine and synthetic cannabinoids (Coopman and Cordonnier, 2017; Armenian et al., 2018), due to their ease of manufacturing and readily available precursors shipped from China (Armenian et al., 2018). In addition to being present in other drugs supply, fentanyl analogs have been also marketed equally "enquiry chemicals" and can easily be acquired over the internet. Due to their high potency and the increased utilize of heroin as an initiating opioid of abuse (8.7% in 2005 vs. 33.3% users in 2015) (Cicero et al., 2017; O'Donnell et al., 2017), the number of opioid-related deaths accept drastically increased in the recent years. Given that opioid novices have limited tolerance to opioids, a slight imprecision in dosing inherent in heroin utilise and/or the presence of potent fentanyl and analogs, can be fatal.
Fentanyl, its analogs (due east.g., acetyl fentanyl, 3-methylfentanyl, alphamethylfentanyl, furanyl fentanyl) and the new generation constructed opioids (eastward.one thousand., AH-7921, U-47700, MT-45) take a chemical core construction totally different from morphine, a naturally occurring opioid from Papaver somniferum and reference compound of the opioids group; but all of them act on the opioid receptor (mu-receptor) reducing the intensity of hurting and showing a high addiction potential. These opioid receptor agonists also induce dose-dependent respiratory depression (Pattinson, 2008), which is the main reason for their life-threatening chance (Ujváry et al., 2017). Fentanyl is approximately 200 times more potent than morphine, and the potencies of its analogs are variable, from 7 times more than potent than morphine for butyrfentanyl and furanyl fentanyl, to more than 4,000 and 10,000 times for sufentanil and carfentanil, respectively (UNODC, 2017). The new generation opioids AH-7921 and MT-45 show similar potency to morphine (Brittain et al., 1977; EMCDDA, 2015), and U-47700 about 7.5 times more strong (Cheney et al., 1985).
Constructed opioids are widely regulated past the United states of america Controlled Substances Act of 1970 (CSA) in gild to control their use and distribution. Equally new compounds arise and threaten public safety, compounds can be emergency scheduled by the DEA to tedious production and use of these harmful substances and aid in prosecution of drug diverters for a temporary menstruum until the formal procedures have gone through (US Drug Enforcement Administration, 2017). Substances are classified into schedules in the CSA based on their safe, medicinal use and potential for corruption. A Schedule I substance is classified as having no currently accustomed medical utilise and a high abuse potential. Examples of synthetic opioids in Schedule I include furanyl fentanyl, U-47700, acetyl fentanyl and 3-methyl fentanyl. Schedule II classified opioids take a high potential for abuse only accept electric current medicinal uses like fentanyl which is used every bit an anesthetic and analgesic, as well as carfentanil, remifentanil and sufentanil (United states Drug Enforcement Administration, 2017). Most recently, the DEA issued a temporary scheduling society for all fentanyl –related substances (to include all analog modifications) in February of 2018, which cover all substances that were not already classified into Schedule I of the CSA in an aggressive attempt to regulate the manufacture and subsequent trafficking of new synthetic opioids into the United States (Drug Enforcement Administration, 2018).
The expansion of these new synthetic opioids constitutes an important claiming in forensic toxicology. Showtime of all, most of these substances are not detected in the routine screening and confirmation methods in the laboratory. As well, due to the depression doses employed of these highly potent drugs, the concentrations expected in the biological samples are in the low ng to pg/mL or ng to pg/g range, requiring extremely sensitive methods of assay. Recently, Marchei et al. (2018) and Liu et al. (2018) reviewed the currently available screening and confirmation methods of new synthetic opioids in biological and not-biological samples. As indicated by Marchei et al. (2018), gas chromatography combined with mass spectrometry (GC-MS) and more frequently liquid chromatography tandem mass spectrometry (LC-MSMS) are the most common techniques due to their sensitivity and specificity. Still, given the continued evolution of new derivatives, the major disadvantage of these target techniques, which apply quadrupole mass spectrometers, is that are limited by the reference standards available. High resolution mass spectrometry (time-of-flying, orbitrap) offers potential advantages to identify unknown compounds without the availability of a reference standard, just this technology is not readily available in most forensic laboratories (Marchei et al., 2018).
Regarding biological samples, most of these methods take been developed in blood or urine, and the target analytes are the parent compounds and rarely the metabolites (Marchei et al., 2018). In postmortem toxicology, other biological specimens such as vitreous humor, liver and brain are commonly analyzed. Unfortunately, fully validated methods for the decision of constructed opioids in these specimens are lacking in the literature. This is in office due to the constant changes in illicit synthetic opioids being identified and laboratories existence unable to justify the extensive time and price associated with fully validating a method for a drug that may merely be present in cases for a brusque fourth dimension. Analytical methods in forensic toxicology are commonly validated in the corresponding biological sample following the guidelines published by the Scientific Working Group in Forensic Toxicology (SWGTOX) (Scientific Working Group for Forensic Toxicology, 2013) to guarantee the belittling quality of the measured concentrations. The analysis of metabolites in the unlike biological matrices may meliorate the interpretation of the results, extending the detection window and indicating if it was an acute or a delayed-death evaluating the metabolite-to-parent ratios. Recent publications nigh the identification of new metabolites of the synthetic opioids are bachelor (Wohlfarth et al., 2016; Steuer et al., 2017; Watanabe et al., 2017; Krotulski et al., 2018a); even so, its application to authentic samples is nonetheless scarce (Poklis et al., 2015; Staeheli et al., 2016; Martucci et al., 2017; Allibe et al., 2018).
Besides the analytical challenges associated with synthetic opioids, due to the scarcity of available postmortem data, the interpretation of the results is extremely difficult. Conducting postmortem toxicology interpretation provides a number of very significant challenges to the forensic toxicologist. The range of postmortem specimens (blood, urine, vitreous sense of humour, tissues, hair), the lack of reference databases, the presence of other substances (e.m., benzodiazepines, alcohol), opioid tolerance, and postmortem phenomena (postmortem redistribution and drug instability) complicates the estimation of the analytical findings. Pichini et al. (2018) and Zawilska (2017) discussed non-fatal and lethal intoxications involving the new synthetic opioids, and Drummer (2018) focused his review on fatalities due to these compounds.
The present review is focused on fentanyl derivatives and new generation opioids due to the limited knowledge concerning these substances and their high prevalence in opioid-overdose related cases. This work complements the previously published literature reviewing the current noesis of postmortem toxicology of constructed opioids and the chemical and pharmacological factors that may affect drug concentrations in the different matrices and therefore, their interpretation in postmortem samples. These factors include central chemical properties, essential pharmacokinetics parameters, postmortem redistribution and stability data in postmortem samples. All of these data are critically compared to postmortem information of natural opioids (morphine), semi-synthetic (oxycodone, hydrocodone, hydromorphone, and oxymorphone), and synthetic opioids (methadone and buprenorphine). The interpretation of drug intoxication in decease investigation is based on the available published literature. This review serves to facilitate the evaluation of cases where synthetic opioids may be implicated in a fatality through the review of peer reviewed published instance reports and research articles.
Methods
PubMed, Scopus and Google Scholar were searched for appropriate articles. Forensic instance-reports and research articles of natural, semi-synthetic and synthetic opioids were reviewed up to May 2018. All articles were manually reviewed for content and references in each manuscript were further queried. Included articles were express to peer-reviewed journals indexed by the Institute for Scientific Information (ISI) and published in English. Chemic properties were retrieved from the public databases PubChem (https://pubchem.ncbi.nlm.nih.gov/) and DrugBank (https://www.drugbank.ca/drugs).
Chemical and Pharmacological Properties
The chemical construction of the diverse synthetic opioids, including fentanyl and analogs, differs significally from the chemical structure of morphine and semi-synthetic opioids (e.g., oxycodone, hydrocodone, buprenorphine). Figure i summarizes the chemical construction of selected classic opioids. Fentanyl is a piperidinyl derivative with moieties on the nitrogen and the iv-position (Figure 2). The different fentanyl derivatives bear witness substitutions on the propionyl moiety (e.g., acetylfentanyl, acrylfentanyl, butyrfentanyl, furanyl fentanyl), phenethyl moiety (e.g., ohmefentanyl), North-phenyl ring (e.g., ocfentanil, iv-methoxy-butyrylfentanyl) and/or at the 4-piperidinyl-position (e.g., carfentanil). The chemical structures of the new generation synthetic opioids (AH-7921, U-47700, MT-45) are unlike from fentanyl. Figure 3 shows 20 fentanyl derivatives and 3 new generation synthetic opioids not related to fentanyl. Due to the close chemical structure among fentanyl derivatives, some compounds, such as cyclopropyl fentanyl and crotonyl fentanyl, take exactly the aforementioned molecular formula, and therefore, the same molecular weight. As a consequence of this, special attending has to be paid in the development of the belittling methods for the determination of these compounds, and a consummate chromatographic separation is required to guarantee their correct identification past gas or liquid chromatography coupled to mass spectrometry (GC-MS, LC-MSMS).
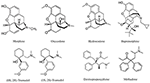
Figure 1. Chemical structures of selected classic opioids.
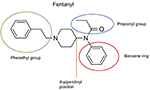
Figure 2. Chemic construction of fentanyl.
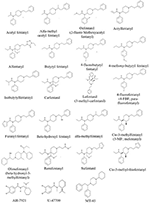
Figure three. Chemical structures of twenty fentanyl derivatives and 3 new generation opioids non related to fentanyl.
Chemically, opioids are predominantly basic drugs with pKa ranging from vii.v to 10.nine. The chemical parameter log P, the decimal logarithm of the partitioning coefficient Kp, is a useful indication of the lipophilicity of a compound. In the case of opioids, log P range is wide, from 0.8 (oxymorphone) to 5 (methadone). Morphine and related compounds show the lowest log P values (0.8–2). Fentanyl and analogs show a log P between 1.5 and four.3. The loftier lipophilicity of fentanyl and its analogs enables rapid diffusion through membranes, including the blood-brain barrier. Also, this lipophilicity forth with their bones characteristics brand these grouping of drugs candidates to undergo postmortem redistribution. Table ane summarizes the molecular weight, pKa and log P of selected opioids.
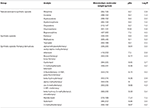
Table ane. Monoisotopic molecular weight (g/mol), pKa and Log P of selected natural, semi-synthetic and synthetic opioids.
Volume of distribution (Vd) and poly peptide binding likewise help to predict the drugs that may exhibit postmortem redistribution. Vd is defined as the volume into which the full amount of the drug would have to be uniformly distributed to reach the concentrations measured in plasma. It is expressed in Fifty/kg of body weight (amount of drug in the body divided by the plasma drug concentration). Drugs highly bound to plasma proteins just not to tissue components would be expected to have a small Vd, while those drugs which distribute into muscle, adipose tissue and other intracellular components will have a loftier Vd. Drugs with a Vd greater than three L/kg are considered to have a greater potential to undergo postmortem redistribution. Table 2 summarizes the Vd and poly peptide binding data currently available for selected opioids.
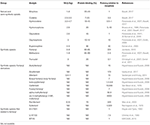
Tabular array two. Critical pharmacological backdrop in postmortem toxicology, book of distributon (Vd), poly peptide bining and authority relative to morphine, of selected natural, semi-synthetic and constructed opioids.
One of the critical problems related to fentanyl, its derivatives and the new synthetic opioids, is the low concentrations expected in the biological samples (ng to pg/mL or ng to pg/yard range) due to their high potency. However, the potency of these type of drugs varies considerably within this group, and therefore the concentrations reported show a broad range, depending on the drug. Tabular array 2 summarizes the potencies relative to morphine for selected opioids.
Metabolism
The identification and quantification of metabolites in postmortem samples may improve the interpretation of the analytical results. The determination of metabolites may extend the window of detection, and also can exist employed to calculate metabolite-to-parent ratios in urine and other biological samples to differentiate acute or delayed death. In certain cases, as information technology happens in morphine and buprenorphine, metabolites can be pharmacologically active. Although this type of information is limited in the case of the synthetic opioids, fentanyl, sufentanil, and alfentanil's metabolites are inactive in the opioid system (Schneider and Brune, 1986).
Although the utility of metabolite determination in biological samples is known, its application to authentic specimens is still deficient in the case of synthetic opioids due to the limited data available nigh their metabolism (Poklis et al., 2015; Staeheli et al., 2016; Martucci et al., 2017; Allibe et al., 2018). Recent publications about the identification of new metabolites of the constructed opioids in vivo and in vitro are available (Wohlfarth et al., 2016; Steuer et al., 2017; Watanabe et al., 2017; Krotulski et al., 2018a). While in vitro studies utilizing man liver hepatocytes or microsomes can identify multiple primary and secondary metabolites for a particular fentanyl derivative, actual man specimens typically show lower number and/or a dissimilar metabolite prevalence profile, and so studies investigating the presence of the in vitro metabolites in authentic homo samples are highly encouraged. Table iii summarizes recent publications about the identification of new metabolites of constructed opioids in vitro and in vivo.

Table 3. In vitro and in vivo metabolism of synthetic opioids.
Fentanyl-derivatives metabolism studies showed similarities and differences from fentanyl metabolism pathways and rates. These different metabolic pathways observed for certain derivatives, demonstrate the need to perform individual metabolism studies for each new compound. In the case of fentanyl, only less than eight% of fentanyl is excreted unchanged. Approximately 85% of the dose is excreted within 72 h in carrion and urine, the majority as metabolites mainly as norfentanyl generated by N-dealkylation at the piperidine nitrogen (McClain and Hug, 1980). Small-scale fentanyl metabolites are despropionylfentanyl, also known equally 4-ANPP, which is formed by carboxamide hydrolysis, and hydroxyfentanyl and hydroxynorfentanyl metabolites, both hydroxylated at the propionyl moiety (Goromaru et al., 1984; Mahlke et al., 2014).
Several synthetic opioids follow a similar metabolic pathway to fentanyl. Alfentanil undergoes piperidine North-dealkylation to noralfentanil (Meuldermans et al., 1988). Major blastoff-methylfentanyl metabolites in rats were norfentanyl and hydroxypropionyl norfentanyl metabolites, exactly as fentanyl (Sato et al., 2010). Meyer et al. (2012) investigated the metabolism in rats of isofentanyl and 3-methyl fentanyl. Later the assistants of suspected recreational doses, the parent drugs could not be detected in urine and their common nor-metabolite was the predominant compound.
Patton et al. (2014) detected high concentrations of acetylfentanyl and acetyl norfentanyl (>16,500 ng/mL, 180 min post-dose) in urine samples from rats treated with a toxic dose of acetylfentanyl (3 mg/kg); however, Melent'ev et al. (2015), showed that the main pathway of the biotransformation of acetylfentanyl was hydroxylation by the phenylethyl moiety rather than North-dealkylation in authentic human samples. Melent'ev et al. (2015) and Watanabe et al. (2017) recommended every bit target analytes in human urine hydroxy-methoxy at phenylethyl moiety and monohydroxylated metabolites, although the reported hydroxylation position in both publications was dissimilar. In both publications, the parent compound acetylfentanyl was highly abundant in urine samples, indicating that the parent drug is a suitable target.
Acrylfentanyl underwent N-dealkylation at the piperidine nitrogen producing the major nor-metabolite (Watanabe et al., 2017). The parent compound was also detected at high concentrations in urine samples. Northward-Dealkylation and monohydroxylation of the piperidine ring were the dominant metabolic pathways for carfentanil in vitro (Feasel et al., 2016). In that written report, the authors observed a slow parent depletion in the hepatocytes. For 4-fluoroisobutyrylfentanyl the main metabolites identified in urine were the nor-metabolite, and monohydroxy metabolites at the piperidine band or at the ethyl linker, besides as the parent chemical compound. In terms of specificity, Watanabe et al., recommended equally target compounds in urine the monohydroxy metabolites and the hydroxymethoxy metabolite (Watanabe et al., 2017).
In the case of butyrfentanyl, hydroxylation of the butanamide side concatenation followed by subsequent oxidation to the carboxylic acid represented the major metabolic footstep (Steuer et al., 2017). Although the norbutyrfentanyl was not among the nigh abundant metabolites in human being samples in that study, the authors suggested its inclusion as a recommended target analyte because it showed a high intensity in the in vitro experiment. In accurate postmortem blood and urine samples, butyrfentanyl was still detected at 66 and 1,000 ng/mL, respectively.
Furanylfentanyl contains a furan group that affects its metabolic profile. This structure seemed to favor the amide hydrolysis, which is the primary metabolite in vitro and in vivo (Watanabe et al., 2017). In terms of specificity of the target metabolites, Watanabe et al. (2017) recommended the dihydrodiol-metabolite and Goggin et al. (2017) recommended the same metabolite, besides every bit the sulfate of the metabolite that results from the amide hydrolysis. As information technology happened with butyrfentanyl (Steuer et al., 2017), the hepatocyte experiment too suggested high prevalence for the nor-metabolite, which was not significantly present in the authentic urine samples, illustrating the need to clarify man specimens. Furanylfentanyl parent compound was detected in accurate urine samples. For ocfentanyl, the predominant metabolite detected in claret, forth with the parent drug, was the O-desmethylated metabolite (Allibe et al., 2018).
In the example of the new synthetic opioids not structurally related to fentanyl, different metabolic pathways has been reported. For AH-7921, the preferred metabolic sites were the amine function and the cyclohexyl band. The 2 nigh dominant metabolites after hepatocyte incubation (also identified in a urine case specimen) were desmethyl and di-desmethyl AH-7921. Together with the glucuronidated metabolites, they were recommended as suitable analytical targets for documenting AH-7921 intake (Wohlfarth et al., 2016). In the case of MT-45, Montesano et al reported hydroxy-MT-45-glucuronide and di-hydroxy-MT-45-glucuronide as the near abundant metabolites in rat urine, while the parent drug was plant at concentrations <10 ng/mL after 300 min (Montesano et al., 2017). Although similar in chemic structure, U-47700 and U-49900 showed specific metabolites. Northward-Desmethyl-U-47700 was identified as the major metabolite in human urine specimens, and Northward,N-Didesethyl-N-desmethyl-U-49900 was identified every bit the about arable metabolite present. Dissimilar U-47700 specimens, U-49900 was detected in low abundance in urine samples (Krotulski et al., 2018a).
As indicated by Watanabe et al. (2017), the target metabolites should generally exist abundant, specific of the parent drug, and prevalent in most, if not all, case samples. Given the strong structural similarities among emerging designer fentanyls, many of them are coincidentally biotransformed to the exact same metabolite. This fact tin make identification of the specific parent drug in a instance difficult. The ability to identify minor metabolites that are unique and specific to the parent drug is therefore of considerable importance. 4-ANPP can exist formed by fentanyl and other different fentanyl analogs metabolism, and it is too a precursor contaminant institute in seized illicit fentanyl and analogs, so its presence is not particularly diagnostic. Other mutual metabolites are: acetylnorfentanyl from acetyl-alpha-methylfentanyl or acetylfentanyl (Watanabe et al., 2017); norfentanyl from fentanyl, beta-hydroxythiofentanyl and alpha-methyl-fentanyl (Sato et al., 2010); norcarfentanil from carfentanil, sufentanil and remifentanil (Feasel et al., 2016). 3,4-dichloro-N-(2-aminocyclohexyl)-N-methyl-benzamide is a common metabolite of U-47700 and U-49900, but it is not a major metabolite in urine for either chemical compound (Krotulski et al., 2018a).
Another of import aspect of the metabolism is the identification of the enzymes involved. Pharmacokinetic interactions may exist produced due to the presence of other substances metabolized by the same enzymes, ultimately affecting the drug blood concentrations. Fentanyl, sufentanyl and alfentanil are mainly metabolized by CYP 3A4 (Feierman and Lasker, 1996; Guitton et al., 1997). Steuer et al., identified CYP 3A4 and CYP 2D6 as the isoforms involved in the metabolism of butyrfentanyl (Steuer et al., 2017). Meyer et al., reported that CYP 3A4, CYP 3A5 and CYP 2C19 are involved in the metabolism of 3-methylfentanyl and isofentanyl and, in the case of isofentanyl, additionally CYP2D6 (Meyer et al., 2012). Remifentanil is the only family unit member of this grade institute to exist ~95% metabolized in the blood and tissues by non-CYP enzymes, probably due to an hands accessible ester group assuasive rapid hydrolysis by circulating blood esterases (Bürkle et al., 1996).
Concentrations in Postmortem Specimens and Other Findings
The concentrations determined in postmortem specimens varied considerably depending on the type of synthetic opioid detected. Derivatives with potencies relative to morphine of more than 170, showed concentrations in femoral blood in the low ng/mL or pg/mL range, while those derivatives with potencies similar to morphine showed concentrations of hundreds, and even thousands, of ng/mL. An exception happens with furanyl fentanyl, which is seven times more potent than morphine (Higashikawa and Suzuki, 2008), just the reported femoral concentrations were less than 50 ng/mL. Typical morphine postmortem concentrations in blood in fatalities are from 200 to 2,300 ng/mL, for methadone 400 to 1,800 ng/mL, for buprenorphine i.ane–29 ng/mL and norbuprenorphine (agile metabolite) 0.2–13 ng/mL (Baselt, 2017), and for oxymorphone 23–554 ng/mL (Crum et al., 2013). The say-so of the dissimilar drugs affects their lethal levels, but other important issues, such equally the presence of other CNS depressant drugs, and developed opioids tolerance, have to be taken into account in the interpretation of the concentrations. The derivative with the highest number of published cases was acrylfetanyl, and with the lowest MT-45. Table 4 summarizes the concentrations of the parent drugs found in case reports and articles where overdose due to a specific opioid was the crusade of death.
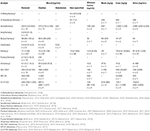
Table four. Postmortem concentrations in dissimilar biological samples for synthetic opioids (median, range, number of cases).
In several cases, multiple constructed opioids were detected. Acetylfentanyl and fentanyl were frequently found together (Pearson et al., 2015; Poklis et al., 2015; Dwyer et al., 2018). Other combinations were butyryl fentanyl and acetyl fentanyl (McIntyre et al., 2016b; Poklis et al., 2016), or U-47700 (Mohr et al., 2016); furanyl fentanyl and acetyl fentanyl (Papsun et al., 2017), acryl fentanyl (Butler et al., 2017), butyryl fentanyl (Mohr et al., 2016), fentanyl (Guerrieri et al., 2017a), or carfentanil (Shanks and Behonick, 2017); carfentanil and fentanyl (Shanks and Behonick, 2017); and tetrahydrofuran fentanyl and U-49900 (Krotulski et al., 2018b). The femoral concentrations reported in those combination cases were often beneath the range of the concentrations summarized in Tabular array 4. Acetylfentanyl median and concentration range in multiple constructed opioids cases were 9.4, 0.4–240 ng/mL (n = fifteen); acrylfentanyl 0.three ng/mL (northward = 1); butyrfentanyl fourteen.9, 0.3–58 ng/mL (northward = four); carfentanil 0.08, 0.05–0.ane ng/mL (n = 2); fentanyl viii.2, 1.1–38 ng/mL (due north = 14); furanyl fentanyl one.seven, 0.6–6.i ng/mL (north = four) and U-47700 17 ng/mL (n = one).
In all of the reports mentioned in Table 4 and in a higher place, synthetic opioids were normally detected with other drugs, specially other CNS depressants, such as benzodiazepines, ethanol and other opioids. This combination may produce a pharmacodynamic interactions and increase the risk of respiratory depression. This possible interaction betwixt opioids, alcohol and benzodiazepines has been previously described for other opioids, such equally buprenorphine (Häkkinen et al., 2012; Seldén et al., 2012), methadone (Jones et al., 2012; Pilgrim et al., 2013; Nielsen et al., 2015), oxycodone (Ogle et al., 2012), and heroin (Thaulow et al., 2014). Amongst the reviewed cases positive for synthetic opioids other than fentanyl, 44 reported every bit cause of death intoxication due to multiple drugs and 77 intoxication mainly due to one specific opioid. The manner of expiry was predominantly accidental (due north = 99), and suicides were reported in seven cases.
Postmortem Redistribution and Stability
Postmortem changes in drug concentrations can happen via postmortem redistribution (PMR) from tissues of a higher to a lower concentration. Physicochemical and pharmacological properties of the analytes, such as pKa, log P, volume of distribution (Vd) and protein bounden, may indicate drugs that feel this postmortem miracle. Lipophilic basic drugs with a Vd > iii L/kg, such as fentanyl, may undergo PMR. Fentanyl has been reported to undergo all-encompassing PMR (Luckenbill et al., 2008; Olson et al., 2010; Palamalai et al., 2013; Brockbals et al., 2018). In the case of the constructed opioids, limited data is currently bachelor about PMR, and as well as information most pKa, log P and Vd (Tables 2, 3). Staeheli et al. (2016) reported postmortem concentration changes of butyrfentanyl and metabolites, suggesting these compounds were prone to PMR. PMR reports about other constructed opioids are not currently available.
Based on currently published case reports and articles, the cardiac blood-to-femoral claret and liver-to-femoral blood ratios were calculated to predict candidates of PMR. Results are summarized in Tabular array 5. Due to the deficient amount of data available (1–4 cases per analyte), no conclusions could be drawn. Synthetic opioids showed median cardiac-to-femoral ratios around 1, and a trend to accrue in the liver. Regarding the distribution to vitreous humor, it may be slow showing higher concentrations in blood. Other factors, such as time of expiry and sample collection, or rapid vs. delayed deaths, has non been taken into business relationship in this analysis due to the limited data bachelor.

Table five. Postmortem concentration ratios in different biological samples for constructed opioids (median, range, number of cases).
PMR is still a controversial issue for classic opioids. Hargrove and Molina (2014) showed insignificant redistribution of morphine from central sites inside 24 h after death in bodies kept at 4°C, while Staeheli et al. (2017) observed a significant increment of morphine concentration, although these changes were not relevant for forensic interpretation. Morphine-derivatives, such us hydrocodone (Saitman et al., 2015), codeine (Frost et al., 2016), and oxycodone (Brockbals et al., 2018), are unlikely to undergo substantial PMR changes. More lipophilic opioids with college Vd, similar methadone (Jantos and Skopp, 2013; Holm and Linnet, 2015; Brockbals et al., 2018), may undergo PMR.
Several studies have been conducted to evaluate stability of fentanyl and some of its derivatives in fortified biological samples, such as claret, plasma and urine. 11 fentanils (fentanyl, norfentanyl, carfentanil, norcarfentanil, sufentanil, norsufentanil, lofentanil, 3-methylfentanyl, alfa-methylfentanyl, ohmefentanyl, and remifentanil acid metabolite), were stable in urine samples stored at −20°C or below for at least two months. However, remifentanil in urine samples decreased by approximately 90% inside 1 calendar week at room temperature and by more than than fifty% in samples stored for ane calendar week at 4°C. Because of the instability of that analyte, the authors recommended to analyze the primary metabolite, remifentanil acid (Wang and Bernert, 2006). Fentanyl and its metabolites norfentanyl, despropionylfentanyl and hydroxynorfentanyl were stable in urine after 3 freeze-thaw cycles, and after storage at −20°C for ii months (Mahlke et al., 2014).
Fentanyl, norfentanyl, acetyl fentanyl and acetyl norfentanyl spiked into whole blood were stable after iii freeze-thaw cycles and at room temperature for 72 h (Poklis et al., 2015). No loss of fentanyl concentration could exist observed later 3 months of storage at 4–8°C and −20°C in blood samples at 5 and 10 ng/mL (Andresen et al., 2012). Nevertheless, another report showed fentanyl and its metabolites norfentanyl, despropionylfentanyl and hydroxynorfentanyl lose up to 51.6% after iii freeze-thaw cycles, and fentanyl and despropionylfentanyl upwards to 34.viii% after storage at −20°C for 2 months (Mahlke et al., 2014). Furanylfentanyl showed no pregnant degradation in claret samples at 5 and 10 ng/mL 48 h room temp and at four°C 7 days (Guerrieri et al., 2017a) and up to thirty days (Mohr et al., 2016).
Regarding the new synthetic opioids not related to fentanyl, U-47700 was stable in blood refrigerated for up to thirty days (Mohr et al., 2016). AH-7921 was found to be stable for at to the lowest degree 21 days in blood and plasma at room temperature (Soh and Elliot, 2014). In the example of MT-45, a loss of 50% was observed afterwards 12 months of storage (Papsun et al., 2016). Farther studies are necessary to evaluate the stability of the different synthetic opioids and metabolites, and in additional biological samples of forensic involvement, such as vitreous humor and tissues.
Conclusion
We performed a critical review of the currently available literature to assist in the toxicological interpretation of constructed opioids postmortem cases. Synthetic opioids plant a heterogenous group of compounds related or not to fentanyl, by and large basic and lipophilic, with a wide range of potencies related to morphine, from one to 10,000. Research has been conducted in the investigation of metabolic pathways and identification of target metabolites of fentanyl derivatives and non-structurally related synthetic opioids, showing similarities and differences from fentanyl depending on the chemical compound. Postmortem concentrations seemed to correlate with their potency, although the presence of other CNS depressants, such equally ethanol and benzodiazepines has to exist taken into account. Farther enquiry is guaranteed to investigate postmortem redistribution phenomena of this grade of compounds, and stability issues in postmortem samples.
Writer Contributions
MC and GC contributed conception and design of the review. MC, RC, and JP searched, organized, reviewed and analyzed the case reports and inquiry articles. MC wrote the commencement typhoon of the manuscript. All authors contributed to manuscript revision, read and canonical the submitted version.
Disharmonize of Involvement Argument
The authors declare that the research was conducted in the absence of whatever commercial or financial relationships that could be construed as a potential conflict of interest.
References
Allibe, N., Richeval, C., Phanithavong, M., Faure, A., Allorge, D., Paysant, F., et al. (2018). Fatality involving ocfentanil documented by identification of metabolites. Drug Exam. Anal. 10, 995–1000. doi: 10.1002/dta.2326
PubMed Abstract | CrossRef Full Text | Google Scholar
Anderson, D. T., and Muto, J. J. (2000). Duragesic transdermal patch: postmortem tissue distribution of fentanyl in 25 cases. J. Anal. Toxicol. 24, 627–634. doi: x.1093/jat/24.vii.627
PubMed Abstract | CrossRef Total Text | Google Scholar
Andresen, H., Gullans, A., Veselinovic, M., Anders, South., Schmoldt, A., Iwersen-Bergmann, Due south., et al. (2012). Fentanyl: toxic or therapeutic? postmortem and antemortem blood concentrations later transdermal fentanyl awarding. J. Anal. Toxicol. 36, 182–194. doi: ten.1093/jat/bks005
PubMed Abstract | CrossRef Full Text | Google Scholar
Armenian, P., Vo, Grand. T., Barr-Walker, J., and Lynch, K. 50. (2018). Fentanyl, fentanyl analogs and novel constructed opioids: a comprehensive review. Neuropharmacology 134, 121–132. doi: 10.1016/j.neuropharm.2017.10.016
PubMed Abstract | CrossRef Total Text | Google Scholar
Baselt, R. C. (2017). Disposition of Toxic Drugs and Chemicals in Man, 11th Edn. Seal Beach, CA: Biomedical Pubns.
Biedrzycki, O. J., Bevan, D., and Lucas, Southward. (2009). Fatal overdose due to prescription fentanyl patches in a patient with sickle cell/β-thalassemia and acute chest syndrome: a case report and review of the literature. Am. J. Forensic Med. Pathol. 30, 188–190. doi: x.1097/PAF.0b013e318187de71
PubMed Abstract | CrossRef Total Text | Google Scholar
Brittain, R. T., Jack, D., and Tyers, M. B. (1977). Pharmacological and certain chemic properties of AH 10407, an unusually short-interim, competitive neuromuscular blocking drug, and some related compounds. Br. J. Pharmacol. 61, 47–55.
PubMed Abstruse | Google Scholar
Brockbals, L., Staeheli, Due south. N., Gascho, D., Ebert, Fifty. C., Kraemer, T., and Steuer, A. E. (2018). Time-dependent postmortem redistribution of opioids in blood and alternative matrices. J. Anal. Toxicol. 42, 365–374. doi: 10.1093/jat/bky017
PubMed Abstract | CrossRef Full Text | Google Scholar
Bruera, E., Pereira, J., Watanabe, S., Belzile, M., Kuehn, N., and Hanson, J. (1996). Opioid rotation in patients with cancer pain. Cancer 78, 852–857. doi: 10.1002/(SICI)1097-0142(19960815)78:four<852::AID-CNCR23>three.0.CO;2-T
PubMed Abstract | CrossRef Full Text | Google Scholar
Butler, D. C., Shanks, 1000., Behonick, K. Southward., Smith, D., Presnell, S. E., and Tormos, L. M. (2017). Iii cases of fatal acrylfentanyl toxicity in the united states and a review of literature. J. Anal. Toxicol. 42, e6–e11. doi: 10.1093/jat/bkx083
PubMed Abstract | CrossRef Full Text | Google Scholar
Carson, H. J., Knight, Fifty. D., Dudley, G. H., and Garg, U. (2010). A fatality involving an unusual route of fentanyl delivery: chewing and aspirating the transdermal patch. Leg. Med. 12, 157–159. doi: 10.1016/j.legalmed.2010.03.001
PubMed Abstract | CrossRef Total Text | Google Scholar
Cheney, B. V., Szmuszkovicz, J., Lahti, R. A., and Zichi, D. A. (1985). Factors affecting binding of trans-N-[two-(Methylamino)cyclohexyl]benzamides at the primary morphine receptor. J. Med. Chem. 28, 1853–1864. doi: 10.1021/jm00150a017
PubMed Abstract | CrossRef Full Text | Google Scholar
Christoph, T., Kögel, B., Strassburger, W., and Schug, S. A. (2007). Tramadol has a better potency ratio relative to morphine in neuropathic than in nociceptive hurting models. Drugs R D 8, 51–57. doi: 10.2165/00126839-200708010-00005
PubMed Abstruse | CrossRef Full Text | Google Scholar
Coopman, V., and Cordonnier, J. (2017). 'Spice-like' herbal incense laced with the synthetic opioid U-47700. Toxicol. Anal. Clin. 30, 75–79. doi: 10.1016/j.toxac.2017.07.004
CrossRef Total Text | Google Scholar
Coopman, V., Cordonnier, J., De Leeuw, M., and Cirimele, Five. (2016). Ocfentanil overdose fatality in the recreational drug scene. Forensic Sci. Int. 266, 469–473. doi: ten.1016/j.forsciint.2016.07.005
PubMed Abstract | CrossRef Full Text | Google Scholar
Coopman, V., Cordonnier, J., Pien, K., and Van Varenbergh, D. (2007). LC-MS/MS analysis of fentanyl and norfentanyl in a fatality due to application of multiple durogesic® transdermal therapeutic systems. Forensic Sci. Int. 169, 223–27. doi: 10.1016/j.forsciint.2006.03.018
CrossRef Full Text | Google Scholar
Crum, E. D., Bailey, K. M., Richards-Waugh, L. L., Clay, D. J., Gebhardt, M. A., and Kraner, J. C. (2013). Validation of blood and liver oxymorphone analysis using LC-MS-MS: concentrations in 30 fatal overdoses. J. Anal. Toxicol. 37, 512–516. doi: 10.1093/jat/bkt077
PubMed Abstruse | CrossRef Full Text | Google Scholar
Dahan, A., Yassen, A., Bijl, H., Romberg, R., Sarton, E., Teppema, L., et al. (2005). Comparison of the respiratory furnishings of intravenous buprenorphine and fentanyl in humans and rats. Br. J. Anaesth. 94, 825–34. doi: ten.1093/bja/aei145
PubMed Abstract | CrossRef Full Text | Google Scholar
Drug Enforcement Administration Department of Justice. (2018). Schedules of Controlled substances: temporary placement of fentanyl related substances in schedule I. Fed. Reg. 83, 5188–5192.
Drummer, O. (2018). Fatalities caused by novel opioids: a review. Forensic Sci. Res. doi: 10.1080/20961790.2018.1460063. [Epub ahead of print].
CrossRef Total Text | Google Scholar
Dussy, F. East., Hangartner, South., Hamberg, C., Berchtold, C., Scherer, U., Schlotterbeck, G., et al. (2016). An acute ocfentanil fatality: a case study with postmortem concentrations. J. Anal. Toxicol. 40, 761–766. doi: ten.1093/jat/bkw096
PubMed Abstract | CrossRef Full Text | Google Scholar
Dwyer, J. B., Janssen, J., Luckasevic, T. Thousand., and Williams, K. E. (2018). Report of increasing overdose deaths that include acetyl fentanyl in multiple counties of the southwestern region of the commonwealth of Pennsylvania in 2015–2016. J. Forensic Sci. 63, 195–200. doi: 10.1111/1556-4029.13517
PubMed Abstract | CrossRef Full Text | Google Scholar
Dziadosz, M., Klintschar, Chiliad., and Teske, J. (2017). Postmortem concentration distribution in fatal cases involving the synthetic opioid U-47700. Int. J. Legal Med. 131, 1555–1556. doi: x.1007/s00414-017-1593-7
PubMed Abstract | CrossRef Full Text | Google Scholar
Elliott, S. P., Brandt, S. D., and Smith, C. (2016). The offset reported fatality associated with the synthetic opioid iii,iv-Dichloro-N-[2-(Dimethylamino)Cyclohexyl]-N-Methylbenzamide (U-47700) and implications for forensic analysis. Drug Examination. Anal. viii, 875–879. doi: 10.1002/dta.1984
CrossRef Full Text | Google Scholar
EMCDDA (2015). Report on the risk cess of MT-45 in the framework of the Council Decision on new psychoactive substances. Grand duchy of luxembourg: Publications Office of the Eu. Available online at: http://www.emcdda.europa.european union/publications/risk-assessments/mt-45_en
Feasel, Yard. G., Wohlfarth, A., Nilles, J. M., Pang, S., Kristovich, R. L., and Huestis, One thousand. A. (2016). Metabolism of carfentanil, an ultra-potent opioid, in human liver microsomes and human hepatocytes by high-resolution mass spectrometry. AAPS J. xviii, 1489–1499. doi: 10.1208/s12248-016-9963-five
PubMed Abstract | CrossRef Full Text | Google Scholar
Feierman, D. E., and Lasker, J. 1000. (1996). Metabolism of fentanyl, a synthetic opioid analgesic, by man liver microsomes. Office of CYP3A4. Drug Metab. Disp. 24, 932–939.
PubMed Abstract | Google Scholar
Fels, H., Krueger, J., Sachs, H., Musshoff, F., Graw, M., Roider, M., et al. (2017). 2 fatalities associated with synthetic opioids: AH-7921 and MT-45. Forensic Sci. Int. 277, e30–e35. doi: x.1016/j.forsciint.2017.04.003
PubMed Abstract | CrossRef Total Text | Google Scholar
Frost, J., Løkken, T. N., Helland, A., Nordrum, I. S., and Slørdal, L. (2016). Postal service-mortem levels and tissue distribution of codeine, codeine-half-dozen-glucuronide, norcodeine, morphine and morphine glucuronides in a series of codeine-related deaths. Forensic Sci. Int. 262, 128–137. doi: 10.1016/j.forsciint.2016.02.051
PubMed Abstract | CrossRef Full Text | Google Scholar
Gaulier, J.-M., Richeval, C., Gicquel, T., Hugbart, C., Le Cartel, B., Allorge, D., et al. (2017). In vitro characterization of nps metabolites produced past human liver microsomes and the hepaRG prison cell line using liquid chromatography-loftier resolution mass spectrometry (LC-HRMS) assay: application to furanyl fentanyl. Curr. Pharm. Biotechnol. 18, 806–814. doi: x.2174/1389201018666171122124401
CrossRef Full Text | Google Scholar
Goggin, M. M., Nguyen, A., and Janis, G. C. (2017). Identification of unique metabolites of the designer opioid furanyl fentanyl. J. Anal. Toxicol. 41, 367–375. doi: 10.1093/jat/bkx022
PubMed Abstract | CrossRef Total Text | Google Scholar
Goromaru, T., Matsuura, H., Yoshimura, N., Miyawaki, T., Sameshima, T., Miyao, J., et al. (1984). Identification and quantitative determination of fentanyl metabolites in patients by gas chromatography-mass spectrometry. Anesthesiology 61, 73–77. doi: 10.1097/00000542-198407000-00013
PubMed Abstract | CrossRef Full Text | Google Scholar
Guerrieri, D., Rapp, E., Roman, M., Druid, H., and Kronstrand, R. (2017a). Postmortem and toxicological findings in a series of furanylfentanyl-related deaths. J. Anal. Toxicol. 41, 242–249. doi: 10.1093/jat/bkw129
PubMed Abstract | CrossRef Full Text | Google Scholar
Guerrieri, D., Rapp, E., Roman, G., Thelander, M., and Kronstrand, R. (2017b). Acrylfentanyl: some other new psychoactive drug with fatal consequences. Forensic Sci. Int. 277, e21–e29. doi: x.1016/j.forsciint.2017.05.010
PubMed Abstract | CrossRef Full Text | Google Scholar
Guitton, J., Desage, Thousand., Alamercery, S., Dutruch, L., Dautraix, Southward., Perdrix, J. P., et al. (1997). Gas chromatographic – mass spectrometry and gas chromatographic – fourier transform infrared spectroscopy assay for the simultaneous identification of fentanyl metabolites. J. Chromatogr. B 59, 59–lxx. doi: 10.1016/S0378-4347(97)00050-9
CrossRef Total Text | Google Scholar
Häkkinen, M., Launiainen, T., Vuori, Eastward., and Ojanperä, I. (2012). Benzodiazepines and booze are associated with cases of fatal buprenorphine poisoning. Eur. J. Clin. Pharmacol. 68, 301–309. doi: 10.1007/s00228-011-1122-4
PubMed Abstruse | CrossRef Full Text | Google Scholar
Hayes, A. Yard., and Tyers, Chiliad. B. (1983). Determination of receptors that mediate opiate side effects in the mouse. Br. J. Pharmacol. 79, 731–736. doi: x.1111/j.1476-5381.1983.tb10011.ten
PubMed Abstract | CrossRef Total Text | Google Scholar
Higashikawa, Y., and Suzuki, S. (2008). Studies on 1-(2-Phenethyl)-4-(North-Propionylanilino)Piperidine (Fentanyl) and its related compounds. VI. structure-analgesic activity relationship for fentanyl, methyl-substituted fentanyls and other analogues. Forensic Toxicol. 26, 1–5. doi: ten.1007/s11419-007-0039-1
CrossRef Full Text | Google Scholar
Hikin, L., Smith, P. R., Ringland, E., Hudson, Due south., and Morley, S. R. (2018). Multiple fatalities in the north of england associated with synthetic fentanyl analogue exposure: detection and quantitation a case serial from early on 2017. Forensic Sci. Int. 282, 179–183. doi: ten.1016/j.forsciint.2017.11.036
PubMed Abstract | CrossRef Total Text | Google Scholar
Holm, Chiliad. M., and Linnet, Grand. (2015). Distribution of enantiomers of methadone and its primary metabolite eddp in human tissues and blood of postmortem cases. J. Forensic Sci. lx, 95–101. doi: 10.1111/1556-4029.12627
PubMed Abstract | CrossRef Full Text | Google Scholar
Jantos, R., and Skopp, Grand. (2013). Postmortem blood and tissue concentrations of R- and S-enantiomers of methadone and its metabolite EDDP. Forensic Sci. Int. 226, 254–60. doi: 10.1016/j.forsciint.2013.01.038
PubMed Abstract | CrossRef Full Text | Google Scholar
Jones, A. West., Holmgren, A., and Ahlner, J. (2012). Claret methadone concentrations in living and deceased persons: variations over time, discipline demographics, and relevance of coingested drugs. J. Anal. Toxicol. 36, 12–eighteen. doi: ten.1093/jat/bkr013
PubMed Abstract | CrossRef Full Text | Google Scholar
Kanamori, T., Togawa Iwata, Y., Segawa, H., Yamamuro, T., Kuwayama, K., Tsujikawa, Thousand., et al. (2018). Metabolism of fentanyl and acetylfentanyl in human-induced pluripotent stalk cell-derived hepatocytes. Biol. Pharm. Bull. 41, 106–114. doi: 10.1248/bpb.b17-00709
PubMed Abstract | CrossRef Total Text | Google Scholar
Karinen, R., Tuv, South. S., Rogde, S., Peres, Yard. D., Johansen, U., Frost, J., et al. (2014). Lethal poisonings with AH-7921 in combination with other substances. Forensic Sci. Int. 244, e21–e24. doi: 10.1016/j.forsciint.2014.08.013
PubMed Abstract | CrossRef Full Text | Google Scholar
Krinsky, C. South., Lathrop, S. L., Crossey, 1000., Baker, G., and Zumwalt, R. (2011). A toxicology-based review of fentanyl-related deaths in new united mexican states (1986–2007). Am. J. Forensic Med. Pathol. 32, 347–351. doi: 10.1097/PAF.0b013e31822ad269
PubMed Abstract | CrossRef Full Text | Google Scholar
Krinsky, C. Due south., Lathrop, S. L., and Zumwalt, R. (2014). An examination of the postmortem redistribution of fentanyl and interlaboratory variability. J. Forensic Sci. 59, 1275–1279. doi: 10.1111/1556-4029.12381
PubMed Abstract | CrossRef Full Text | Google Scholar
Kronstrand, R., Thelander, G., Lindstedt, D., Roman, M., and Kugelberg, F. C. (2014). Fatal intoxications associated with the designer opioid AH-7921. J. Anal. Toxicol. 38, 599–604. doi: 10.1093/jat/bku057
PubMed Abstract | CrossRef Total Text | Google Scholar
Krotulski, A. J., Mohr, A. L. A., Papsun, D. M., and Logan, B. K. (2018a). Metabolism of novel opioid agonists U-47700 and U-49900 using human liver microsomes with confirmation in authentic urine specimens from drug users. Drug Test. Anal. 10, 127–136. doi: x.1002/dta.2228
PubMed Abstract | CrossRef Full Text | Google Scholar
Krotulski, A. J., Papsun, D. M., Friscia, Thou., Swartz, J. Fifty., Holsey, B. D., and Logan, B. K. (2018b). Fatality post-obit ingestion of tetrahydrofuranylfentanyl, U-49900 and Methoxy-Phencyclidine. J. Anal. Toxicol. 42, e27–32. doi: 10.1093/jat/bkx092
PubMed Abstract | CrossRef Full Text | Google Scholar
Kuhlman, J. J., McCaulley, R., Valouch, T. J., and Behonick, M. South. (2003). Fentanyl use, misuse, and abuse: a summary of 23 postmortem cases. J. Anal. Toxicol. 27, 499–504. doi: 10.1093/jat/27.7.499
PubMed Abstract | CrossRef Total Text | Google Scholar
Liu, Fifty., Wheeler, Due south. E., Venkataramanan, R., Rymer, J. A., Pizon, A. F., Lynch, Chiliad. J., et al. (2018). Newly emerging drugs of abuse and their detection methods. Am. J. Clin. Pathol. 149, 105–116. doi: 10.1093/ajcp/aqx138
PubMed Abstract | CrossRef Total Text | Google Scholar
Luckenbill, K., Thompson, J., Middleton, O., Kloss, J., and Apple, F. (2008). Fentanyl postmortem redistribution: preliminary findings regarding the relationship amidst femoral claret and liver and heart tissue concentrations. J. Anal. Toxicol. 32, 639–643. doi: 10.1093/jat/32.8.639
PubMed Abstract | CrossRef Full Text | Google Scholar
Mahlke, North. S., Ziesenitz, Five., Mikus, G., and Skopp, G. (2014). Quantitative depression-book assay for simultaneous conclusion of fentanyl, norfentanyl, and small metabolites in human plasma and urine past liquid chromatography—tandem mass spectrometry (LC-MS/MS). Int. J. Legal Med. 128, 771–778. doi: 10.1007/s00414-014-1040-y
PubMed Abstract | CrossRef Total Text | Google Scholar
Marchei, E., Pacifici, R., Mannocchi, G., Marinelli, E., Busardò, F. P., and Pichini, Due south. (2018). New constructed opioids in biological and non-biological matrices: a review of electric current analytical methods. Trends Anal. Chem. 102, 1–15. doi: 10.1016/j.trac.2018.01.007
CrossRef Full Text | Google Scholar
Marinetti, L. J., and Ehlers, B. J. (2014). A serial of forensic toxicology and drug seizure cases involving illicit fentanyl alone and in combination with heroin, cocaine or heroin and cocaine. J. Anal. Toxicol. 38, 592–598. doi: 10.1093/jat/bku086
CrossRef Total Text | Google Scholar
Martin, T. L., Woodall, K. L., and McLellan, B. A. (2006). Fentanyl-related deaths in ontario, canada: toxicological findings and circumstances of decease in 112 cases (2002-2004). J. Anal. Toxicol. thirty, 603–610. doi: 10.1093/jat/30.8.603
PubMed Abstract | CrossRef Total Text | Google Scholar
Martucci, H. F. H., Ingle, East. A., Hunter, M. D., and Rodda, 50. N. (2017). Distribution of furanyl fentanyl and iv-ANPP in an accidental acute expiry: a case report. Forensic Sci. Int. 283, e13–e17. doi: 10.1016/j.forsciint.2017.12.005
PubMed Abstract | CrossRef Full Text | Google Scholar
McIntyre, I. G., Gary, R. D., Estrada, J., and Nelson, C. L. (2014). Antemortem and postmortem fentanyl concentrations: a case written report. Int. J. Legal Med. 128, 65–67. doi: x.1007/s00414-013-0897-5
PubMed Abstract | CrossRef Full Text
McIntyre, I. M., Trochta, A., Gary, R. D., Malamatos, Grand., and Lucas, J. R. (2016a). An acute acetyl fentanyl fatality: a case report with postmortem concentrations. J. Anal. Toxicol. 40:88. doi: 10.1093/jat/bkv043
CrossRef Total Text
McIntyre, I. Yard., Trochta, A., Gary, R. D., Wright, J., and Mena, O. (2016b). An acute butyrfentanyl fatality: a case report with postmortem concentrations. J. Anal. Toxicol. forty, 162–166. doi: 10.1093/jat/bkv138
CrossRef Full Text | Google Scholar
Melent'ev, A. B., Kataev, S. S., and Dvorskaya, O. Due north. (2015). Identification and belittling backdrop of acetyl fentanyl metabolites. J. Anal. Chem. seventy, 240–248. doi: x.1134/S1061934815020124
CrossRef Full Text | Google Scholar
Meuldermans, Due west., Van Peer, A., Hendrickx, J., Woestenborghs, R., Lauwers, W., Heykants, J., et al. (1988). Alfentanil pharmacokinetics and metabolism in humans. Anesthesiology 69, 527–534. doi: 10.1097/00000542-198810000-00012
PubMed Abstract | CrossRef Full Text | Google Scholar
Meyer, M. R., Dinger, J., Schwaninger, A. E., Wissenbach, D. Yard., Zapp, J., Fritschi, Grand., et al. (2012). Qualitative studies on the metabolism and the toxicological detection of the fentanyl-derived designer drugs 3-methylfentanyl and isofentanyl in rats using liquid chromatography-linear ion trap-mass spectrometry (LC-MS N). Anal. Bioanal. Chem. 402, 1249–1255. doi: 10.1007/s00216-011-5528-8
PubMed Abstract | CrossRef Full Text | Google Scholar
Mohr, A. L., Friscia, M., Papsun, D., Kacinko, S. L., Buzby, D., and Logan, B. K. (2016). Analysis of novel synthetic opioids U-47700, U-50488 and furanyl fentanyl by LC-MS/MS in postmortem casework. J. Anal. Toxicol. 40, 709–717. doi: 10.1093/jat/bkw086
PubMed Abstract | CrossRef Full Text | Google Scholar
Montesano, C., Vannutelli, G., Fanti, F., Vincenti, F., Gregori, A., Rita Togna, A., et al. (2017). Identification of MT-45 metabolites: in silico prediction, in vitro incubation with rat hepatocytes and in vivo confirmation. J. Anal. Toxicol. 41, 688–697. doi: 10.1093/jat/bkx058
PubMed Abstract | CrossRef Full Text | Google Scholar
Moore, P. Due west., Palmer, R. B., and Donovan, J. W. (2015). Fatal fentanyl patch misuse in a hospitalized patient with a postmortem increment in fentanyl claret concentration. J. Forensic Sci. 60, 243–246. doi: 10.1111/1556-4029.12559
PubMed Abstract | CrossRef Full Text | Google Scholar
Nielsen, Yard. Thou., Johansen, S. Due south., and Linnet, K. (2015). Evaluation of poly-drug apply in methadone-related fatalities using segmental hair analysis. Forensic Sci. Int. 248, 134–139. doi: 10.1016/j.forsciint.2015.01.004
PubMed Abstract | CrossRef Full Text | Google Scholar
Niemegeers, C. J., Schellekens, Yard. H., Van Bever, Westward. F., and Janssen, P. A. (1976). Sufentanil, a very potent and extremely rubber intravenous morphine-like compound in mice, rats and dogs. Arzneimittelforschung 26, 1551–1556.
PubMed Abstract | Google Scholar
O'Donnell, J. K., Halpin, J., Mattson, C. L., Goldberger, B. A., and Gladden, R. K. (2017). Deaths Involving Fentanyl, Fentanyl Analogs, and U-47700 — 10 States, July–December 2016. MMWMorbidity R Mortal. Weekly Report 66, 1197–1202.
PubMed Abstruse
Oertel, R., Pietsch, J., Arenz, N., Zeitz, S. G., Goltz, 50., and Kirch, W. (2011). Distribution of metoprolol, tramadol, and midazolam in human autopsy material. J. Chromatogr. A 1218, 4988–94. doi: x.1016/j.blush.2010.12.113
PubMed Abstract | CrossRef Total Text | Google Scholar
Ogle, A., Moore, M., Barrett, B., Young, Chiliad. S., and Pearson, J. (2012). Clinical history and characteristics of persons with oxycodone-related deaths in hillsborough county, Florida in 2009. Forensic Sci. Int. 223, 47–52. doi: 10.1016/j.forsciint.2012.07.016
PubMed Abstruse | CrossRef Full Text | Google Scholar
Ojanperä, I., Gergov, M., Rasanen, I., Lunetta, P., Toivonen, S., Tiainen, Due east., et al. (2006). Blood levels of iii-methylfentanyl in 3 fatal poisoning cases. American J. Forensic Med. Pathol. 27, 328–331. doi: 10.1097/01.paf.0000188097.78132.e5
PubMed Abstract | CrossRef Full Text
Olson, K. N., Luckenbill, K., Thompson, J., Middleton, O., Geiselhart, R., Mills, One thousand. M., et al. (2010). Postmortem redistribution of fentanyl in blood. Am. J. Clin. Pathol. 133, 447–453. doi: 10.1309/AJCP4X5VHFSOERFT
PubMed Abstract | CrossRef Full Text | Google Scholar
Palamalai, V., Olson, Yard. North., Kloss, J., Middleton, O., Mills, K., Strobl, A. Q., et al. (2013). Superiority of postmortem liver fentanyl concentrations over peripheral claret influenced by postmortem interval for decision of fentanyl toxicity. Clin. Biochem. 46, 598–602. doi: 10.1016/j.clinbiochem.2013.02.001
PubMed Abstract | CrossRef Full Text | Google Scholar
Papsun, D., Hawes, A., Mohr, A. L. A., Friscia, M., and Logan, B. G. (2017). Case serial of novel illicit opioid-related deaths. Acad. Forensic Pathol. 7, 477–486. doi: 10.23907/2017.040
CrossRef Full Text | Google Scholar
Papsun, D., Krywanczyk, A., Vose, J. C., Bundock, East. A., and Logan, B. One thousand. (2016). Analysis of MT-45, a novel synthetic opioid, in human whole blood by LC-MS-MS and its identification in a drug-related death. J. Anal. Toxicol. 40, 313–317. doi: x.1093/jat/bkw012
PubMed Abstract | CrossRef Full Text | Google Scholar
Patton, A. L., Seely, K. A., Pulla, S., Rusch, North. J., Moran, C. Fifty., Fantegrossi, W. Eastward., et al. (2014). Quantitative measurement of acetyl fentanyl and acetyl norfentanyl in human urine past LC-MS/MS. Anal. Chem. 86, 1760–1766. doi: 10.1021/ac4036197
PubMed Abstract | CrossRef Full Text | Google Scholar
Pearson, J., Poklis, J., Poklis, A., Wolf, C., Mainland, M., Hair, 50., et al. (2015). Postmortem toxicology findings of acetyl fentanyl, fentanyl, and morphine in heroin fatalities in tampa, Florida. Acad. Forensic Pathol. 5, 676–689. doi: 10.23907/2015.072
PubMed Abstract | CrossRef Full Text | Google Scholar
Pichini, S., Solimini, R., Berretta, P., Pacifici, R., and Busardò, F. P. (2018). Acute intoxications and fatalities from illicit fentanyl and analogues. Ther. Drug Monit. 40, 38–51. doi: 10.1097/FTD.0000000000000465
PubMed Abstract | CrossRef Total Text | Google Scholar
Pilgrim, J. 50., McDonough, K., and Drummer, O. H. (2013). A review of methadone deaths between 2001 and 2005 in Victoria, Australia. Forensic Sci. Int. 226, 216–22. doi: 10.1016/j.forsciint.2013.01.028
PubMed Abstract | CrossRef Full Text | Google Scholar
Poklis, J., Poklis, A., Wolf, C., Hathaway, C., Arbefeville, Due east., Chrostowski, L., et al. (2016). Two fatal intoxications involving butyryl fentanyl. J. Anal. Toxicol. 40, 703–708. doi: x.1093/jat/bkw048
PubMed Abstract | CrossRef Full Text | Google Scholar
Poklis, J., Poklis, A., Wolf, C., Mainland, M., Pilus, L., Devers, K., et al. (2015). Postmortem tissue distribution of acetyl fentanyl, fentanyl and their corresponding nor-metabolites analyzed by ultrahigh functioning liquid chromatography with tandem mass spectrometry. Forensic Sci. Int. 257, 435–441. doi: 10.1016/j.forsciint.2015.10.021
PubMed Abstract | CrossRef Full Text | Google Scholar
Rodda, L. Due north., Pilgrim, J. Fifty., Di Rago, G., Crump, K., Gerostamoulos, D., and Drummer, O. H. (2017). A cluster of fentanyl-laced heroin deaths in 2015 in Melbourne, Commonwealth of australia. J. Anal. Toxicol. 41, 318–324. doi: 10.1093/jat/bkx013
PubMed Abstract | CrossRef Full Text | Google Scholar
Rohrig, T. P., Miller, Southward. A., and Baird, T. R. (2017). U-47700: a non so new opioid. J. Anal. Toxicol. 42, e12–e14. doi: 10.1093/jat/bkx081
CrossRef Full Text | Google Scholar
Rojkiewicz, K., Majchrzak, Chiliad., Celinski, R., Kuś, P., and Sajewicz, G. (2017). Identification and physicochemical characterization of 4-fluorobutyrfentanyl (ane-((four-Fluorophenyl)(1-Phenethylpiperidin-4-Yl)Amino)Butan-1-One, 4-FBF) in seized materials and post-mortem biological samples. Drug Exam. Anal. 9, 405–414. doi: 10.1002/dta.2135
PubMed Abstract | CrossRef Full Text | Google Scholar
Saitman, A., Fitzgerald, R. Fifty., and McIntyre, I. M. (2015). Evaluation and comparison of postmortem hydrocodone concentrations in peripheral blood, fundamental blood and liver specimens: a minimal potential for redistribution. Forensic Sci. Int. 247, 36–forty. doi: 10.1016/j.forsciint.2014.eleven.031
PubMed Abstract | CrossRef Full Text | Google Scholar
Sato, S., Suzuki, Southward., Lee, X. P., and Sato, K. (2010). Studies on 1-(ii-Phenethyl)-4-(N-Propionylanilino)Piperidine (Fentanyl) and related compounds. vii. quantification of α-methylfentanyl metabolites excreted in rat urine. Forensic Sci. Int. 195, 68–72. doi: x.1016/j.forsciint.2009.11.014
PubMed Abstract | CrossRef Total Text | Google Scholar
Schneider, E., and Brune, Chiliad. (1986). Opioid activity and distribution of fentanyl metabolites. Naunyn Schmiedeberg'south Curvation. Pharmacol. 334, 267–274.
PubMed Abstract | Google Scholar
Scientific Working Group for Forensic Toxicology (2013). Scientific Working Group for Forensic Toxicology (SWGTOX) standard practices for method validation in forensic toxicology. J. Anal. Toxicol. 37, 452–474. doi: 10.1093/jat/bkt054
CrossRef Full Text
Seldén, T., Ahlner, J., Druid, H., and Kronstrand, R. (2012). Toxicological and pathological findings in a series of buprenorphine related deaths. possible take chances factors for fatal consequence. Forensic Sci. Int. 220, 284–ninety. doi: 10.1016/j.forsciint.2012.03.016
PubMed Abstruse | CrossRef Full Text | Google Scholar
Seth, P., Scholl, L., Rudd, R. A., and Bacon, S. (2018). Overdose deaths involving opioids, cocaine, and psychostimulants — United States, 2015 – 2016. MMWR Morb. Mortal. Wkly. Rep. 67, 349–358 doi: x.15585/mmwr.mm6712a1
PubMed Abstract | CrossRef Full Text | Google Scholar
Shanks, Thousand. G., and Behonick, Thou. S. (2017). Detection of carfentanil by LC-MS-MS and reports of associated fatalities in the Usa. J. Anal. Toxicol. 41, 466–472. doi: ten.1093/jat/bkx042
PubMed Abstract | CrossRef Full Text | Google Scholar
Smith, H. S. (2009). Clinical pharmacology of oxymorphone. Pain Med. 10(Suppl. 1), S1–S10. doi: 10.1111/j.1526-4637.2009.00594.x
CrossRef Full Text | Google Scholar
Staeheli, Due south. Northward., Baumgartner, M. R., Gauthier, S., Gascho, D., Jarmer, J., Kraemer, T., et al. (2016). Time-dependent postmortem redistribution of butyrfentanyl and its metabolites in blood and alternative matrices in a case of butyrfentanyl intoxication. Forensic Sci. Int. 266, 170–177. doi: ten.1016/j.forsciint.2016.05.034
PubMed Abstract | CrossRef Full Text | Google Scholar
Staeheli, S. Northward., Gascho, D., Ebert, L. C., Kraemer, T., and Steuer, A. Eastward. (2017). Time-dependent postmortem redistribution of morphine and its metabolites in blood and alternative matrices—application of ct-guided biopsy sampling. Int. J. Legal Med. 131, 379–389. doi: 10.1007/s00414-016-1485-2
PubMed Abstruse | CrossRef Full Text | Google Scholar
Steuer, A. East., Williner, East., Staeheli, S. N., and Kraemer, T. (2017). Studies on the metabolism of the fentanyl- derived designer drug butyrfentanyl in human being in vitro liver preparations and authentic man samples using liquid chromatography-high resolution mass spectrometry (LC-HRMS). Drug Exam. Anal. 9, 1085–1092. doi: x.1002/dta.2111
PubMed Abstract | CrossRef Full Text | Google Scholar
Swanson, D. Grand., Hair, L. S., Strauch Rivers, S. R., Smyth, B. C., Brogan, S. C., Ventoso, A. D., et al. (2017). Fatalities involving carfentanil and furanyl fentanyl: two instance reports. J. Anal. Toxicol. 41, 498–502. doi: 10.1093/jat/bkx037
PubMed Abstract | CrossRef Full Text | Google Scholar
Takase, I., Koizumi, T., Fujimoto, I., Yanai, A., and Fujimiya, T. (2016). An autopsy instance of acetyl fentanyl intoxication caused by insufflation of 'Designer Drugs.' Legal Med. 21, 38–44. doi: 10.1016/j.legalmed.2016.05.006
PubMed Abstract | CrossRef Full Text | Google Scholar
Thaulow, C. H., Høiseth, G., Andersen, J. M., Handal, 1000., and Mørland, J. (2014). Pharmacokinetic interactions between ethanol and heroin: a study on post-mortem cases. Forensic Sci. Int. 242, 127–134. doi: 10.1016/j.forsciint.2014.06.032
PubMed Abstract | CrossRef Full Text | Google Scholar
Ujváry, I., Jorge, R., Christie, R., Le Ruez, T., Danielsson, H. V., Kronstrand, R., et al. (2017). Acryloylfentanyl, a recently emerged new psychoactive substance: a comprehensive review. Forensic Toxicol. 35, 232–243. doi: 10.1007/s11419-017-0367-8
CrossRef Full Text | Google Scholar
UNODC (2017). Recommended Methods for the Identification and Analysis of Fentanyl and Its Analogues in Biological Specimens (Vienna).
Van Bever, W. F., Niemegeers, C. J., Schellekens, Chiliad. H., and Janssen, P. A. (1976). Northward-4-Substituted one-(2-Arylethyl)-4-Piperidinyl-N-phenylpropanamides, a novel series of extremely potent analgesics with unusually high safety margin. Arzneimittelforschung. 26, 1548–1551.
PubMed Abstract | Google Scholar
Vardanyan, R. S., and Hruby, V. J. (2014). Fentanyl-related compounds and derivatives: curresnt status time to come prospects for pharmaceutical applications. Future Med. Chem. vi, 385–412. doi: 10.4155/fmc.13.215
PubMed Abstract | CrossRef Full Text | Google Scholar
Vorce, S. P., Knittel, J. L., Holler, J. M., Magluilo, J., Levine, B., Berran, P., et al. (2014). A fatality involving Ah-7921. J. Anal. Toxicol. 38, 226–230. doi: 10.1093/jat/bku011
PubMed Abstract | CrossRef Full Text | Google Scholar
Wang, 50., and Bernert, J. T. (2006). Analysis of 13 fentanils, including sufentanil and caffentanii, in human urine by liquid ionization-tandem mass spectrometry *. J. Anal. Toxicol. thirty, 335–341.
Google Scholar
Watanabe, Southward., Vikingsson, Southward., Roman, M., Light-green, H., Kronstrand, R., and Wohlfarth, A. (2017). In vitro and in vivo metabolite identification studies for the new synthetic opioids acetylfentanyl, acrylfentanyl, furanylfentanyl, and 4-fluoro-isobutyrylfentanyl. AAPS J. nineteen, 1102–1122. doi: 10.1208/s12248-017-0070-z
PubMed Abstract | CrossRef Full Text | Google Scholar
Wohlfarth, A., Scheidweiler, Yard. B., Pang, S., Zhu, M., Castaneto, Grand., Kronstrand, R., et al. (2016). Metabolic label of AH-7921, a synthetic opioid designer drug: in vitro metabolic stability cess and metabolite identification, evaluation of in silico prediction, and in vivo confirmation. Drug Exam. Anal. eight, 779–791. doi: ten.1002/dta.1856
PubMed Abstruse | CrossRef Total Text | Google Scholar
Yonemitsu, One thousand., Sasao, A., Mishima, Due south., Ohtsu, Y., and Nishitani, Y. (2016). A fatal poisoning instance by intravenous injection of 'Bath Salts' containing acetyl fentanyl and 4-methoxy PV8. Forensic Sci. Int. 267, e6–e9. doi: x.1016/j.forsciint.2016.08.025
PubMed Abstruse | CrossRef Total Text | Google Scholar
Source: https://www.frontiersin.org/articles/10.3389/fphar.2018.01210/full
Post a Comment for "How to Read an Autopsy Toxicology Report Brain"